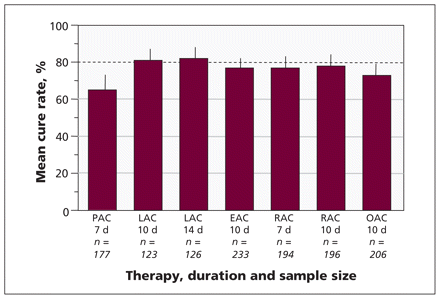
Typically, when choosing antimicrobial therapy for a common infectious disease, clinicians have current information regarding the local level of antibiotic resistance and are able to start therapy empirically while awaiting the results of culture. Infections with Helicobacter pylori are an exception to this rule, in that local antimicrobial resistance patterns are generally unknown, and clinicians must therefore rely entirely on empiric antimicrobial therapy. The most widely recommended anti–H. pylori regimen has been a combination of a proton pump inhibitor and two of amoxicillin, clarithromycin, and metronidazole or tinidazole (known as “legacy triple therapy”). Initially, this was a highly successful regimen, but worldwide resistance to clarithromycin has been increasing, such that treatment success is now generally 80% or less (Figure 1).1–4 In this article, we discuss the choice of anti–H. pylori therapies in this era of increasing antimicrobial resistance.
Results of studies with more than 100 patients that tested the combination of a proton pump inhibitor (first letter in each designation; see below) plus amoxicillin (A) and clarithromycin (C) in North America. The dashed horizontal line signifies the threshold for an acceptable result. The results are shown as mean cure rates (intention-to-treat analysis) and 95% confidence intervals. The number of patients in each study is provided. Note: P = pantoprazole, L = lansoprazole, E = esomeprazole, R = rabeprazole, O = omeprazole. Adapted by permission from BMJ Publishing Group Limited. Gut, Graham DY, Lu H, Yamaoka Y, et al. Vol. 12, p. 275–8, © 2007.4
Regimens capable of producing greater than 90% eradication of H. pylori are available. All of the regimens shown in Box 1 will achieve this high level of success in the treatment of infections with susceptible organisms.1,5 The best advice for clinicians is to prescribe only what is known to work locally and to routinely perform noninvasive post-treatment testing to confirm cure and to determine if resistance has begun to undermine the current locally effective regimens.1 Factors that will assist the clinician in the choice of regimen include the patient’s history of antibiotic use, allergies, and the cost and availability of the drugs.
Recommended regimens for anti–Helicobacter pylori therapy
Empiric therapy*
Concomitant therapy: Combination of four drugs, specifically amoxicillin 1 g, clarithromycin 500 mg, tinidazole or metronidazole 500 mg and a proton pump inhibitor, all given twice daily for 10–14 days. A nitroimidazole twice daily may be added to a legacy triple therapy dose pack if available.
Sequential therapy: Amoxicillin 1 g and a proton pump inhibitor twice daily for five days, then clarithromycin 500 mg, tinidazole or metronidazole 500 mg and a proton pump inhibitor, all twice daily for five days (total 10 days of therapy).
Hybrid (sequential and concomitant) therapy: Amoxicillin 1 g and a proton pump inhibitor twice daily for seven days, then amoxicillin 1 g, a proton pump inhibitor, clarithromycin 500 mg and tinidazole or metronidazole 500 mg, all twice daily for seven days (total 14 days of therapy).
Bismuth quadruple therapy: Bismuth subsalicylate or subcitrate two tabs four times daily, tetracycline 500 mg four times daily (with meals and at bedtime), metronidazole or tinidazole 500 mg three times daily (with meals) and a proton pump inhibitor twice daily for 10 or, preferably, 14 days.
Therapy if results of pretreatment susceptibility testing known
Legacy triple therapy: Three drugs, consisting of two of amoxicillin 1 g, clarithromycin 500 mg, and tinidazole or metronidazole 500 mg, plus a proton pump inhibitor, all given twice daily for 14 days.
Fluoroquinolone-containing triple therapy: Fluoroquinolone once daily, a proton pump inhibitor twice daily and amoxicillin 1 g twice daily for 14 days.
Empiric salvage therapy in cases of multiple treatment failure
High-dose dual therapy with a proton pump inhibitor: Choice of proton pump inhibitor (e.g., omeprazole 40 mg or lansoprazole 30 mg) plus amoxicillin 500 mg four times daily (e.g., at 7 am, 12:30 pm, 7 pm, 11 pm) for 14 days.
Rifabutin-containing triple therapy: Three drugs, consisting of amoxicillin 1 g, rifabutin 150 mg and a proton pump inhibitor, all given twice daily for 14 days.
-
↵* Choice dependent on history of prior antibiotic use and known interactions with or allergies to the medications.
Resistance to clarithromycin and fluoroquinolones (which may also be used in anti–H. pylori therapies) develops rapidly and cannot be overcome by increasing the dose or duration of therapy.1,6,7 In contrast, resistance to metronidazole or tinidazole can be partially overcome by increasing the dose and duration of therapy and by adding a proton pump inhibitor.1,7,8 As a general rule, unless susceptibility testing is available or it is known that legacy triple therapy is locally effective, the clinician should assume that the patient is infected with a clarithromycin-resistant strain of H. pylori and should avoid legacy triple therapy. Similarly, any prior use of a fluoroquinolone (e.g., ciprofloxacin) makes fluoroquinolone resistance highly likely. Resistance to amoxicillin and tetracycline is uncommon,1,7 and prior use of these drugs does not preclude reuse.
Currently recommended regimens
Current successful empiric anti–H. pylori regimens consist of multidrug combinations typically containing four drugs. They can be divided into non-bismuth-containing and bismuth-containing therapies (Box 1).
The effective non-bismuth-containing quadruple regimens use amoxicillin, clarithromycin, metronidazole or tinidazole, and a proton pump inhibitor, in a variety of combinations (Box 1). The first quadruple non-bismuth-containing regimen used these drugs concomitantly, typically twice a day.9 Sequential therapy was a modification in which amoxicillin and a proton pump inhibitor were given for five days, followed by clarithromycin, a nitroimidazole and the proton pump inhibitor for five days (two drugs + three drugs).10,11 In the presence of modest levels of clarithromycin resistance, these regimens proved more successful than legacy triple therapy, generally providing between 90% and 94% eradication.10–12 However, the effectiveness of sequential therapy is reduced, to about 75%, in the face of clarithromycin resistance. As such, effectiveness declines progressively as the prevalence of resistance increases above 20% and likely becomes unacceptably low at about 30% resistance. Despite the large number of individuals who have received sequential therapy, few studies have assessed resistance with these regimens, which makes accurate predictions impossible. The efficacy of sequential regimens is markedly lower in the presence of dual resistance to clarithromycin and metronidazole. In the only head-to-head comparison of sequential and concomitant therapies that has been reported,13 they were equivalent, and the effect of clarithromycin resistance is therefore expected to be similar for these two types of regimen.
Concomitant therapy is less complex than sequential therapy and is our personal choice when deciding between the two types of regimen. However, a new non-bismuth-containing quadruple regimen has been developed that holds considerable promise. This regimen is a hybrid between sequential and concomitant therapy in which the amoxicillin in the initial dual-therapy phase is continued into the second phase (two drugs + four drugs). When given for 14 days (i.e., seven days + seven days), the eradication rate per protocol was 99.1% (95% confidence interval 97.3%–100%, with 117 patients treated).5 Theoretically, hybrid therapy should be superior to both sequential and concomitant therapy in the presence of clarithromycin resistance, as patients with clarithromycin resistance still receive a triple therapy (amoxicillin, metronidazole or tinidazole, and a proton pump inhibitor) rather than only the metronidazole or tinidazole and proton pump inhibitor given in sequential therapy. Additional studies are needed to confirm whether this success holds up in populations with different patterns of antimicrobial resistance.
Bismuth quadruple therapy is a highly successful regimen when the clinician pays attention to the dosing, drugs and duration (Box 1). The most successful bismuth quadruple therapy contains a bismuth salt plus tetracycline, metronidazole or tinidazole, and a proton pump inhibitor.8 There have been no head-to-head comparisons between bismuth subsalicylate and bismuth subcitrate, nor have there been comparisons between different doses of tetracycline (i.e., 375 mg v. 500 mg). However, we have observed that the treatment arms in which H. pylori is most reliably eradicated with this quadruple regimen are those using 10–14 days of therapy, including 20 mg of omeprazole twice a day and at least 1000 mg of both tetracycline and metronidazole or tinidazole.8 As a general rule, we recommend using higher doses of antibiotics four times per day and a 14-day duration for all regimens, unless lower doses and shorter durations have been shown to provide more than 90% (preferably at least 95%) eradication locally.
Treatment failure
If there is failure of eradication with initial therapy or the infection recurs after initial treatment success, the clinician will ideally request culture and susceptibility testing to guide the choice of subsequent therapy. Common causes of initial eradication failure are antimicrobial resistance and poor compliance in taking the drugs as prescribed. Other causes are incorrect choice of drugs (e.g., doxycycline instead of tetracycline), incorrect dosing and insufficient duration of therapy. Resistance can often be addressed with a form of salvage therapy that does not contain any of the drugs used in the initial regimen. As noted above, clarithromycin and fluoroquinolones should not be used a second time, as resistance is likely to develop with use. Metronidazole or tinidazole can be reused, provided it is given at a high dose (1500 or 1600 mg/d, in divided doses at least four times per day) for 14 days. Fluoroquinolones (e.g., moxifloxacin, levofloxacin or, if available, sitafloxacin) are good choices if the patient has never used quinolones (an increasingly rare situation). Generally, we recommend that the clinician switch from a primary quadruple clarithromycin-containing regimen to a salvage bismuth-containing quadruple regimen or vice versa. In each case, it is important to use optimum dosing and duration to increase the probability of treatment success.
Multiple treatment failure represents a special problem and should generally prompt referral to a physician with a special interest in H. pylori therapy. The specialist should carefully review prior drug use and, if susceptibility testing is not available, choose from drugs to which resistance is uncommon (e.g., bismuth, tetracycline, amoxicillin). Furazolidone or rifabutin may also be considered. If there has been no prior use of fluoroquinolones, a fluoroquinolone (e.g., levofloxacin) may be considered, as a component of either triple or sequential salvage therapy1,8,12 (Box 1).
-
Only antimicrobial regimens that have been proven to work locally should be used.
-
After initial treatment failure with clarithromycin or a fluoroquinolone, these drugs should not be reused.
-
Unless low doses are proven to be equally effective locally, full doses of antimicrobials should be used for 14 days.
-
For patients who have experienced multiple treatment failures, treatment recommendations (i.e., salvage therapy) should be sought from an expert in eradication of Helicobacter pylori.
Key points
Footnotes
-
This article was solicited and has been peer reviewed.
-
Competing interests: Dr. Graham is an unpaid consultant for Novartis in relation to vaccine development for the treatment or prevention of infection with Helicobacter pylori. He has served as a paid consultant for Otsuka Pharmaceuticals, and until July 2007 was a board member for Meretek Diagnostics, the manufacturer of the carbon 13 (13C) urea breath test. He has received royalties for the Baylor College of Medicine patent covering materials related to the 13C-labelled urea breath test. He has also provided expert testimony on behalf of Canadian law firms regarding use of proton pump inhibitors. Dr. Fischbach has applied for funding for an investigator-initiated clinical trial comparing the bismuth-containing combination product Pylera (Axcan Pharma) with a new treatment.
-
Contributors: The authors contributed equally to conception and design of the article; acquisition, analysis and interpretation of the data; and drafting and revision of the article for important intellectual concent. Both provided final approval of the version to be published.
-
Funding: This article is based upon work supported in part by the Office of Research and Development, Medical Research Service, US Department of Veterans Affairs. Dr. Graham is supported in part by US Public Health Service grant DK56338, which funds the Digestive Diseases Center within the Texas Medical Center, and also by grants DK067366 and CA116845 from the US National Institutes of Health. The contents are solely the responsibility of the authors and do not necessarily represent the official views of Veterans Affairs or the National Institutes of Health.