Abstract
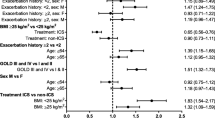
Much controversy surrounds the use of β-agonists in obstructive lung disease. Regular β2-agonist use in asthma results in tolerance to its effects and an increase in asthma-related deaths. Less is known about clinical outcomes in chronic obstructive pulmonary disease (COPD). This systematic review and meta-analysis evaluates the long-term effect of β2-agonist use on severe exacebations requiring hospitalization or trial with drawal, respiratory deaths, and total mortality in patients with COPD. Results for β2-agonists are compared with results for anticholinergics and inhaled corticosteroids. Pooled results from randomized controlled trials show that anticholinergics, such as tiotropium and ipratropium, significantly reduce severe exacerbations and respiratory deaths compared with placebo. Conversely, β2-agonists increase respiratory deaths, probably because of tolerance that develops to their bronchodilator and bronchoprotective effects. Anticholinergics significantly reduce exacerbations and total mortality compared with β-agonists. The combination of the two bronchodilators is not more effective than anticholinergics alone in improving long-term clinical outcomes. Inhaled corticosteroids significantly reduce severe exacerbations and the decline in lung function over time, without affecting mortality. In conclusion, inhaled anticholinergic bronchodilators and corticosteroids should be used to improve long-term clinical outcomes in patients with COPD. β-Agonists increase respiratory deaths in COPD, possibly as a result of poorer disease control.
Similar content being viewed by others
References
Kelman, G. R., Palmer, K.N., and Cross, M. R. (1969), Cardiovascular effects of AH.3365 (salbutamol). Nature 221, 1251.
Dodds, W. N., Soler, N. G., and Thompson, H. (1975), Deaths in asthma. BMJ 4, 345.
Crompton, G. K. (1975), Deaths in asthma. BMJ, 4, 458.
Fahy, J. V., and Boushey, H. A. (1995), Controversies involving inhaled beta-agonists and inhaled corticosteroids in the treatment of asthma. Clin. Chest Med. 16, 715–733.
Taylor, D. R., Sears, M., and Cockcroft, D. W. (1996), The beta-agonist controversy. Med. Clin. N. Am. 80, 719–748.
Lipworth, B. J. (1992), Risks versus benefits of inhaled beta 2-agonists in the management of asthma. Drug Saf. 7, 54–70.
U.S. Food and Drug Administration Advisory Committee. (2005), Serevent, Advair, Foradil withdrawals to be considered by Advisory Committee. http://www.fdaadvisorycommittee.com/FDC/ Advisory Committee/Committees/Pulmonary-Allergy% 20 Drugs/071305_betasafety/071305_BroncoP.htm, Accessed August 8, 2005.
Suissa, S., Blais, L., and Ernst, P. (1994), Patterns of increasing beta-agonist use and the risk of fatal or near-fatal asthma. Eur. Respir. J. 7, 1602–1609.
Au, D. H., Curtis, J. R., and Psaty, B. M. (2001), Risk of myocardial ischaemia and beta-adrenoceptor agonists. Ann. Med. 33, 287–290.
Salpeter, S. R., Ormiston, T. M., and Salpeter, E. E. (2004), Meta-analysis: respiratory tolerance to regular beta2-agonist use in patients with asthma. Ann. Intern. Med. 140, 802–813.
Kraan, J., Koeter, G. H., vd Mark, T. W., Sluiter, H. J., and de Vries, K. (1985), Changes in bronchial hyperreactivity induced by 4 weeks of treatment with antiasthmatic drugs in patients with allergic ashtma: a comparison between budesonide and terbutaline. J. Allergy Clin. Immunol. 76, 628–636.
Sears, M. R., Taylor, D. R., Print, C. G., et al. (1990), Regular inhaled beta-agonist treatment in bronchial asthma. Lancet 336, 1391–1396.
Wahedna, I., Wong, C. S., Wisniewski, A. F., Pavord, I. D., and Tattersfield, A. E. (1993), Asthma control during and after cessation of regular beta 2-agonist treatment. Am. Rev. Respir. Dis. 148, 707–712.
Johnson, M. (1998), The beta-adrenoceptor. Am. J. Respir. Crit. Care Med. 158, S146-S153.
Salpeter, S. R., Ormiston, T. M., and Salpeter, E. E. (2004), Cardiovascular effects of beta-agonists in patients with asthma and COPD: a meta-analysis. Chest 125, 2309–2321.
Salpeter, S. R. (2004). Cardiovascular safety of beta(2)-adrenoceptor agonist use in patients with obstructive airway disease: a systematic review. Drugs Aging 21, 405–414.
U.S. Food and Drug Administration. (2003), Labeling changes for drug products that contain salmeterol. FDA Talk Paper. www.fda.gov/bbs/topics/ANSWERS/2003/ANS01248.html, Accessed August 7, 2005.
Salpeter, S. R., Buckley, N. S., Ormiston, T. M., and Salpeter, E. E. (2006), Meta-analysis: effect of longacting β-agonists on severe asthma exacerbations and asthma-related deaths. Ann. Intern Med. 144, 904–912.
Stelfox, H. T., Chua, G., O'Rourke, K., and Detsky, A. S. (1998), Conflict of interest in the debate over calcium-channel antagonists. N. Engl. J. Med. 338, 101–106.
Bekelman, J. E., Li, Y., and Gross, C. P. (2003), Scope and impact of financial conflicts of interest in biomedical research: a systematic review. JAMA 289, 454–465.
Salpeter, S. R., Buckley, N. S., and Salpeter, E. E. (2006), Meta-analysis: anticholinergics, but not β-agonists, reduce severe exacerbations and respiratory mortality in COPD., J. Gen. Intern. Med. 23, DOI: 10.1111/m. 1525–1497. 2006. 00507. x.
Singh, J. M., Palda, V. A., Stanbrook, M. B., and Chapman, K. R. (2002), Corticosteroid therapy for patients with acute exacerbations of chronic obstructive pulmonary disease: a systematic review. Arch. Intern. Med. 162, 2527–2536.
Gan, W. Q., Man, S. F., and Sin, D. D. (2005), Effects of inhaled corticosteroids on sputum cell counts in stable chronic obstructive pulmonary disease a systematic review and a meta-analysis. BMC Pulm. Med. 5, 3.
Appleton, S., Poole, P., Smith, B., Veale, A., and Bara, A. (2002), Long-acting beta2-agonists for chronic obstructive pulmonary disease patients with poorly reversible airflow limitation. Cochrane Database Syst. Rev. CD001104.
Sestini, P., Renzoni, E. Robinson, S., Poole, P., and Ram, F. S. (2002) Short-acting beta 2 agonists for stable chronic obstructive pulmonary disease. Cochrane Database Syst. Rev. CD001495.
Nannini, L., Cates, C. J., Lasserson, T. J., and Poole, P. (2004), Combined corticosteroid and long acting beta-agonist in one inhaler for chronic obstructive pulmonary disease. Cochrane Database Syst. Rev. CD003794.
McCrory, D. C. and Brown, C. D., (2001), Inhaled short-acting beta2-agonists versus ipratropium for acute exacerbations of chronic obstructive pulmonary disease. Cochrane Database Syst. Rev. CD002984.
Husereau, D., Shukla, V., Boucher, M., Mensinkai, S., and Dales, R. (2004). Long acting beta2 agonists for stable chronic obstructive pulmonary disease with poor reversibility: a systematic review of randomised controlled trials. BMC Pulm. Med. 4, 7.
Keam, S. J., and Keating, G. M. (2004), Tiotropium bromide. A review of its use as maintenance therapy in patients with COPD. Treat/Respir. Med. 3, 247–268.
Barr, R. G., Bourbeau, J., Camargo, C. A., and Ram, F. S. (2005), Inhaled tiotropium for stable chronic obstructive pulmonary disease. Cochrane Database Syst. Rev. CD002876.
Mantel, N. and Haenszel, W. (1959), Statistical aspects of the analysis of data from retrospective studies of disease. J. Natl. Cancer Inst. 22, 719–748.
Celli, B. R., and MacNee, W. (2004), Standards for the diagnosis and treatment of patients with COPD: a summary of the ATS/ERS position paper. Eur. Respir. J. 23, 932–946.
Agusti, A. G. (2005), COPD, a multicomponent disease: implications for management. Respir. Med. 99, 670–682.
Sin, D. D., Lacy, P., York, E., and Man, S. F. (2004), Effects of fluticasone on systemic markers of inflammation in chronic obstructive pulmonary disease. Am. J. Respir. Crit. Care Med. 170, 760–765.
Alsaeedi, A., Sin, D. D., and McAlister, F. A. (2002), The effects of inhaled corticosteroids in chronic obstructive pulmonary disease: a systematic review of randomized placebo-controlled trials. Am. J. Med. 113, 59–65.
van Grunsven, P. M., van Schayck, C. P., Derenne, J. P., et al. (1999), Long term effects of inhaled corticosteroids in chronic obstructive pulmonary disease: a meta-analysis. Thorax 54, 7–14.
Sutherland, E. R., Allmers, H., Ayas, N. T., Venn, A. J., and Martin, R. J. (2003). Inhaled corticosteroids reduce the progression of airflow limitation in chronic obstructive pulmonary disease: a meta-analysis. Thorax 58, 937–941.
Donohue, J. F., Menjoge, S., and Kesten, S. (2003), Tolerance to bronchodilating effects of salmeterol in COPD. Respir. Med. 97, 1014–1020.
Barnes, P. J. (2004), The role of anticholinergics in chronic obstructive pulmonary disease. Am. J. Med. 117 (Suppl 12A), 24S-32S.
National Collaborating Centre for Chronic Conditions. (2004), Chronic obstructive pulmonary disease. National clinical guideline on management of chronic obstructive pulmonary disease in adults in primary and secondary care. Thorax 59 (Suppl 1), 1–232.
Cooper, C. B., and Tashkin D. P. (2005), Recent developments in inhaled therapy in stable chronic obstructive pulmonary disease. BMJ 330, 640–644.
Ramsey, S. D. (2000), Suboptimal medical therapy in COPD: exploring the causes and consequences. Chest 117, 33S-37S.
Rudolf, M. (2000), The reality of drug use in COPD: the European perspective. Chest 117, 29S-32S.
Roche, N., Lepage, T., Bourcereau, J., and Terrioux, P. (2001), Gurdelines versus clinical practice in the treatment of chronic obstructive pulmonary disease. Eur. Respir. J. 18, 903–908.
Aalbers, R., Ayres, J., Backer, V., et al. (2002), Formoterol in patients with chronic obstructive pulmonary disease: a randomized, controlled, 3-month trial. Eur. Respir. J. 19, 936–943.
Bourbeau, J., Rouleau, M. Y., and Boucher, S. (1998), Randomised controlled trial of inhaled corticosteroids in patients with chronic obstructive pulmonary disease. Thorax 53, 477–482.
Boyd, G., Morice, A. H., Pounsford, J. C., Siebert, M., Peslis, N., and Crawford, C. (1997), An evaluation of salmeterol in the treatment of chronic obstructive pulmonary disease (COPD). Eur. Respir. J. 10, 815–821.
Brusasco, V., Hodder, R., Miravitlles, M., Korducki, L., Towse, L., and Kesten, S. (2003), Health outcomes following treatment for six months with once daily tiotropium compared with twice daily salmeterol in patients with COPD. Thorax 58, 399–404.
Burge, P. S., Calverley, P. M., Jones, P. W., Spencer, S., Anderson, J. A., and Maslen, T. K. (2000). Randomised, double blind, placebo controlled study of fluticasone propionate in patients with moderate to severe chronic obstructive pulmonary disease: the ISOLDE trial. BMJ 320, 1297–1303.
Calverley, P. M., Boonsawat, W., Cseke, Z., Zhong, N., Peterson, S., and Olsson, H. (2003). Maintenance therapy with budesonide and formoterol in chronic obstructive pulmonary disease. Eur. Respir. J. 22, 912–919.
Casaburi, R., Briggs, D. D., Jr., Donohue, J. E., Serby, C. W., Menjoge, S. S., and Witek, T. J. Jr. (2000), The spirometric efficacy of once-daily dosing with tiotropium in stable COPD: a 13-week multicenter trial. The US Tiotropium Study Group. Chest 118, 1294–1302.
Casaburi, R., Mahler, D. A., Jones, P. W., et al. (2002), A long-term evaluation of once-daily inhaled tiotropium in chronic obstructive pulmonary disease. Eur. Respir. J. 19, 217–224.
Chapman, K. R., Arvidsson, P., Chuchalin, A. G., et al. (2002), The addition of salmeterol 50 microg bid to anticholinergic treatment in patients with COPD: a randomized, placebo controlled trial Chronic obstructive pulmonary disease. Can. Respir. J. 9, 178–185.
Colice, G. L. (1996), Nebulized bronchodialators for outpatient management of stable chronic obstructive pulmonary disease. Am. J. Med. 100, 11S-18S.
Combivent Inhalation Solution Study Group. (1997), Routine nebulized ipratropium and albuterol together are better than either alone in COPD. The COMBIVENT Inhalation Solution Study Group. Chest 112, 1514–1521.
Cook, D., Guyatt, G., Wong, E., et al. (2001), Regular versus as-needed short-acting inhaled, beta-agonist therapy for chronic obstructive pulmonary disease. Am. J. Respir. Crit. Care Med 163, 85–90.
Donohue, J. F., van Noord, J. A., Bateman, E. D., et al. (2002), A6-month, placebo-controlled study comparing lung function and health status changes in COPD patients treated with tiotropium or salmeterol. Chest 122, 47–55.
Friedman, M., Serby, C. W., Menjoge S. S., Wilson, J. D., Hilleman, D. E., and Witek, T. J. Jr. (1999), Pharmacoeconomic evaluation of a combination of ipratropium plus albuterol compared with ipratropium alone and albuterol alone in COPD. Chest 115, 635–641.
Lung Health Study Research Group. (2000), Effect of inhaled triamcinolone on the decline in pulmonary function in chronic obstructive pulmonary disease. N. Engl. J. Med. 343, 1902–1909.
Mahler, D. A., Wire, P., Horstman, D., et al. (2002), Effectiveness of fluticasone propionate and salmeterol combination delivered via the Diskus device in the treatment of chronic obstructive pulmonary disease. Am. J. Respir. Crit. Care Med. 166, 1084–1091.
Mirici, A., Bektas, Y., Ozbakis, G., and Erman, Z. (2001) Effect of inhaled corticosteroids on respiratory function tests and airway inflammation in stable chornic obstructive pulmonary disease. A randomised, double-blind, placebo-controlled clinical trial. Clin. Drug Invest. 21, 835–842.
Niewoehner, D. E., Rice, K., Cote, C., et al. (2005), Prevention of exacerbations of chronic obstructive pulmonary disease with tiotropium, a one-daily inhaled anticholinergic bronchodilator: a randomized trial. Ann. Intern. Med. 143, 317–326.
Paggiaro, P. L., Dahle, R., Bakran, I., Frith, L., Hollingworth, K., and Efthimiou, J. (1998), Multicentre randomised placebo-controlled trial of inhaled fluticasone propionate in patients with chronic obstructive pulmonary disease. International COPD Study Group. Lancet 351, 773–780.
Pauwels, R. A., Lofdahl, C. G., Laitinen, L. A., et al. (1999), Long-term treatment with inhaled budesonide in persons with mild chronic obstructive pulmonary disease who continue smoking. European Respiratory Society Study on Chronic Obstructive Pulmonary Disease. N. Engl. J. Med. 340, 1948–1953.
Renkema, T. E., Schouten, J. P., Koeter, G. H., and Postma, D. S. (1996), Effects of long-term treatment with corticosteroids in COPD. Chest 109, 1156–1162.
Rennard, S. I., Anderson, W., ZuWallack, R., et al. (2001), Use of a long-acting inhaled beta2-adrenergic agonist, salmeterol xinafoate, in patients with chronic obstructive pulmonary disease. Am. J. Respir. Crit. Care Med. 163, 1087–1092.
Rossi, A., Kristufek, P., Levine, B. E., et al. (2002), Comparison of the efficacy, tolerability, and safety of formoterol dry powder and oral, slow-release theophylline in the treatment of COPD. Chest 121, 1058–1069.
Rutten-van Molken, M., Roos, B., and Van Noord, J. A. (1999), An empirical comparison of the St George's Respiratory Questionnaire (SGRQ) and the Chronic Respiratory Disease Questionnaire (CRQ) in a clinical trial setting. Thorax 54, 995–1003.
Szafranski, W., Cukier, A., Ramirez, A., et al. (2003), Efficacy and safety of budesonide/formoterol in the management of chronic obstructive pulmonary disease. Eur. Respir. J. 21, 74–81.
Tashkin, D. P., Ashutosh, K., Bleecker, E. R., et al. (1986), Comparison of the anticholinergic bronchodilator ipratropium bromide with metaproterenol in chronic obstructive pulmonary disease. A 90-day multi-center study. Am. J. Med. 81, 81–90.
Taylor, J., Kotch, A., Rice, K., et al. (2001), Ipratropium bromide hydrofluoroalkane inhalation aerosol is safe and effective in patients with COPD. Chest 120, 1253–1261.
U.S. Food and Drug Administration. (2002), NDA21-395 Clinical briefing document. Spiriva (Tiotropium bromide) inhalation powder for COPD. http:// www.fda.gov/ohrms/dockets/ac/02/briefing/ 3890B1_05_Clinical%20Briefing-%20Part%201.pdf, Accessed August 10, 2005.
van Noord, J. A., de Munck, D. R., Bantje, T. A., Hop, W. C., Akveld, M. L., and Bommer, A. M. (2000), Long-term treatment of chronic obstructive pulmonary disease with salmeterol and the additive effect of ipratropium. Eur. Respir. J. 15, 878–885.
Vestbo, J., Sorensen, T., Lange, P., Brix, A., Torre, P., and Viskum, K. (1999), Long-term effect of inhaled budesonide in mild and moderate chronic obstructive pulmonary disease: a randomised controlled trial. Lancet 353, 1819–1823.
Wadbo, M., Lofdahl, C. G., Larsson, K., Skoogh, B. E., et al. (2002), Effects of formoterol and ipratropium bromide in COPD: a 3-month placebo-controlled study. Eur. Respir. J. 20, 1138–1146.
Weir, D. C., Bale, G. A., Bright, P., and Sherwood Burge, P. (1999), A double-blind placebo-controlled study of the effect of inhaled beclomethasone dipropionate for 2 years in patients with nonasthmatic chronic obstructive pulmonary disease. Clin. Exp. Allergy 29 (Suppl 2), 125–128.
Ashutosh, K. and Lang, H. (1984), Comparison between long-term treatment of chronic bronchitic airway obstruction with ipratropium bromide and metaproterenol. Ann. Allergy 53, 401–406.
van Schayck, C. P., Graafsma, S. J., Visch, M. B., Dompeling, E., van Weel, C., and van Herwaarden, C. L. (1990), Increased bronchial hyperresponsiveness after inhaling salbutamol during 1 year is not caused by subsensitization to salbutamol. J. Allergy Clin. Immunol. 86, 793–800.
College Ter Beoordeling Van Geneesmiddelen Medicines Evaluation Board. (2002), Public Assessment Report: Spiriva 18 micrograms inhalation powder in hard capsules RVG 26191. http:// www.cbg-meb.nl/nl/docs/gnsmiddl/par-spir iva.pdf.Accessed August 10, 2005.
Vincken, W., van Noord, J. A., Greefhorst, A. P., et al. (2002), Improved health outcomes in patients with COPD during 1 yr's treatment with tiotropium. Eur. Respir. J. 19, 209–216.
Oostenbrink, J. B., Rutten-van Molken, M. P., Al, M. J., Van Noord, J. A., and Vincken, W. (2004). One-year cost-effectiveness of tiotropium versus ipratropium to treat chronic obstructive pulmonary disease. Eur. Respir. J. 23, 241–249.
Gan, W. Q., Man, S. F., Senthilselvan, A., and Sin, D. D. (2004), Association between chronic obstructive pulmonary disease and systemic inflammation: a systematic review and a meta-analysis. Thorax 59, 574–580.
Wood-Baker, R., Walters, E. H., and Gibson, P. (2001), Oral corticosteroids for acute exacerbations of chronic obstructive pulmonary disease. Cochrane Database Syst. Rev. CD001288.
O'Donnell, D. E., Aaron, S., Bourbeau, J., et al. (2003), Canadian Thoracic Society recommendations for management of chronic obstructive pulmonary disease—2003. Can. Respir. J. 10 (Suppl A), 11A-65A.
Pauwels, R. A., Buist, A. S., Calverley, P. M., Jenkins, C. R., and Hurd, S. S. (2001), Global strategy for the diagnosis, management, and prevention of chronic obstructive pulmonary disease. NHLBI/WHO Global Initiative for Chronic Obstructive Lung Disease (GOLD) Workshop summary. Am. J. Respir. Crit. Care Med. 163, 1256–1276.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Salpeter, S.R., Buckley, N.S. Systematic review of clinical outcomes in chronic obstructive pulmonary disease. Clinic Rev Allerg Immunol 31, 219–230 (2006). https://doi.org/10.1385/CRIAI:31:2:219
Issue Date:
DOI: https://doi.org/10.1385/CRIAI:31:2:219